 
Even so, it is already clear that it can not only give us a better understanding of existing substances, but also unleash untapped potential in the elements, promising a whole new world of materials for applications that include solar cells, drugs, data storage and more besides.Īs far back as the 1860s, scientists had begun writing out compounds in a distinctive format with sticks joining element symbols: H-H for the molecule (H 2) made up of two hydrogen atoms, for example. We are still getting to grips with this new panoply of bonding varieties. 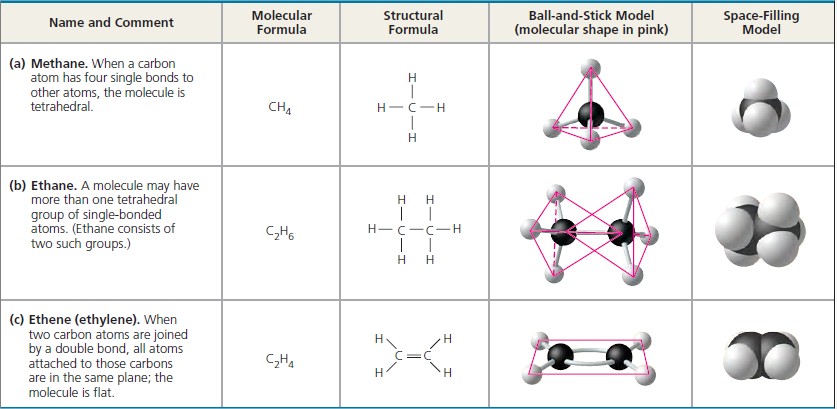
As we have seen, there are two types of bonds: ionic. There are even bonds that, completely against chemical orthodoxy, involve no electrons at all. A molecule or compound is made when two or more atoms form a chemical bond that links them together. Recent discoveries show that there are more types of bond than we thought, and that some of the familiar ones might not be quite as we had imagined. These bonds are a sort of glue that hold the atoms together within the. That makes it all the more shocking that, a century and a half after the idea of chemical bonds was first floated, we are still a long way from a complete understanding of how atoms’ outermost electrons, the mediators of chemical bonds, form these links. New compounds are formed when the atoms within the molecule form a chemical bond. “We talk about chemical bonding because we want to understand and predict materials’ properties,” says Matthias Wuttig, a materials physicist at RWTH Aachen University, Germany. Bonds like this are in oxygen gas nitrogen gas and hydrogen gas. 
When two atoms share electrons between them they are locked together (bonded) by that sharing. And for good reason, because bonds are the glue that binds chemistry together. A chemical bond is a lasting attraction between atoms, ions or molecules that enables the formation of chemical compounds. Atoms come together to form molecules because of their electrons. NO SCHOOL chemistry textbook is complete without a detailed enumeration of the basic types of chemical bond: covalent, ionic and metallic. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
